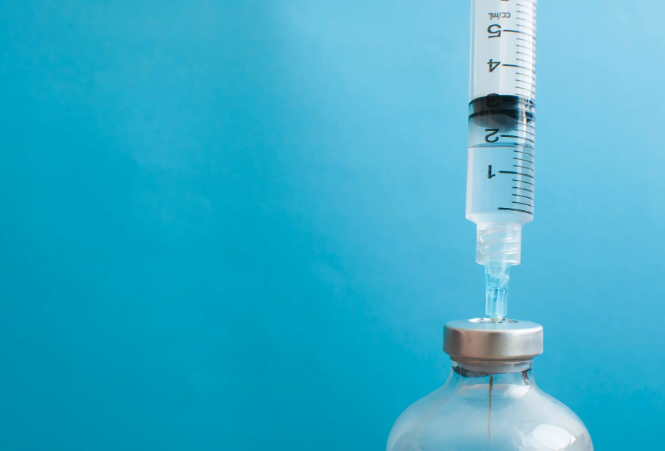
A newly appointed advisory panel at the U.S. Centers for Disease Control and Prevention (CDC) has voted to recommend removing thimerosal, a mercury-based preservative, from flu vaccines—a decision that has sparked concern among public health experts.
The recommendation, passed on June 26 by the Advisory Committee on Immunization Practices (ACIP), now awaits approval by the CDC’s acting director. The move follows sweeping changes within the committee by U.S. Health Secretary Robert F. Kennedy Jr., who replaced all 17 previous members with eight new appointees, some of whom have expressed skepticism about vaccine safety in the past.
Thimerosal, which contains ethylmercury, has been used since the 1930s to prevent bacterial contamination in multi-dose vaccine vials. While most flu vaccines in the U.S. now come in thimerosal-free single-dose syringes, the preservative is still present in about 3-4% of flu vaccines used in rural and low-resource areas due to cost and storage considerations.
Numerous studies over the past two decades have found no evidence that thimerosal causes harm in the low doses used in vaccines. Yet, it has remained a frequent target of anti-vaccine activists. “The thimerosal question has been asked and answered multiple times,” said Dr. Jason Goldman, president of the American College of Physicians. “Now, instead of a fringe group refusing to accept evidence, they are the decision-makers.”
Dr. Cody Meissner, a pediatrician and the only committee member to vote against the recommendation, questioned the rationale behind the move. “Of all the issues ACIP needs to focus on, this is not a big issue,” he told fellow members during the meeting.
Concerns about the legitimacy of the panel have prompted boycotts by several medical groups. The American Academy of Pediatrics (AAP), among others, declined to attend the latest meeting in protest over the removal of the original committee. “We made the decision that this was an illegitimate meeting,” said Dr. Sean O’Leary, chair of the AAP’s infectious diseases committee.
Critics also raised alarm over the CDC’s temporary removal of web pages containing research on thimerosal’s safety just before the committee convened. “This is a clear effort to shine a light on the anti-vaccine trope that thimerosal is somehow dangerous,” O’Leary added.
The timeline for implementation remains unclear. Manufacturers, including Sanofi Pasteur, have stated that thimerosal is already used in only a small fraction of their doses. Still, public health officials warn the decision could strain flu vaccine supplies in underserved areas ahead of the upcoming season.
“If vaccines are no longer appropriately recommended, it will lead to more infections, more deaths, and erode public trust,” said Goldman.